Drug nanoparticles – a common approach to tackle the poor aqueous solubility of APIs
Drug nanoparticles, commonly referred as nanocrystals, are particles made of the drug itself.
A variety of oral dosage forms incorporating drug nanoparticles is marketed (e.g. fenofibrate, sirolimus, aprepitant, megestrol acetate..).
Nanosizing drug particles provides enormous increases in their specific surface area and hence brings key benefits for oral formulations of poorly soluble drugs such as Biopharmaceutics Classification System (BCS) Class II drugs
Improve
Dissolution rate
Bioavailability
Onset of action
Reduce
Pharmacokinetics variability
Dose
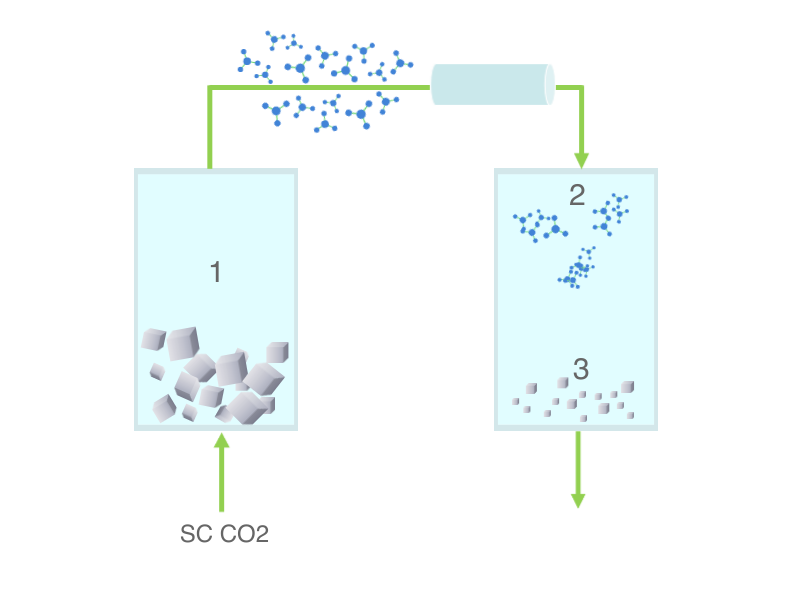
Manufacturing of drug nanoparticles: top-down and solvent-based bottom-up processes
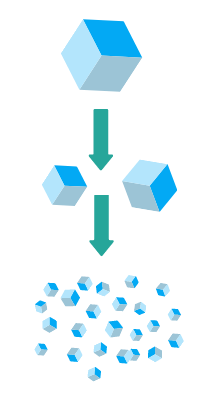
Top-down processes, mainly wet ball milling and high pressure homogenization, are based on size reduction of drug particles dispersed in aqueous vehicles and lead to the recovery of a nanosuspension.
While several marketed products are produced using these processes, the top-down approach has drawbacks which can hinder its application to some challenging APIs:
- poor control over particle size and properties,
- polymorphic transition and/or amorphization
- strict crystallinity specifications of the starting APIs are required for crystallinity batch-to-batch consistency of drug nanoparticles,
- tedious downstream processing to convert the aqueous nanosuspension to a solid dosage form
With bottom-up processes, the drug is dissolved in an organic solvent and the solution is mixed with water and stabilizers to produce a stable nanosuspension.
Bottom-up processes might make it possible to produce nanosuspensions with tailored size.
Nevertheless, some features of these organic solvent based processes often limit their application to pharmaceutical products.
- poor control over crystallinity – amorphous or metastable semi-crystalline particles are often produced
- tedious transformation into solid products as nanoparticles are recovered as a dispersion in an aqueous-organic medium