Polymer and Lipids
High-purity tailored materials for demanding applications
Supercritical fluid technology has proven success in the purification and fractionation of polymers and lipids.
We extend its application to challenging medical and pharmaceutical products.
Supercritical purification and fractionation use no organic solvents, are operated at moderate temperatures and offer unique and tunable selectivity for:
- removal of monomers, oligomers, by-products, organic solvents
- fractionation by molecular weight and side chain branching
A few examples of purification challenges we have solved for our clients:
- Removal of acrylate monomer, synthesis side-products and organic solvents in a medical copolymer
- Purification of a novel lipid used in vaccine formulations
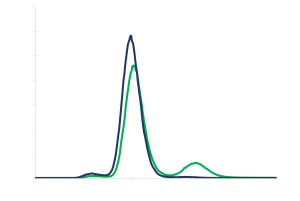
- Fractionation of a pharmaceutical polymer: manufacturing of narrow Mw fractions
- Extraction of residual cyclic siloxanes in silicone polymers
- Cleaning of medical devices: removal of oils, waxes, lubricants
- Fractionation of glycolipids and sphingolipids
- Deoiling of a natural raw material prior to isolation of active proteins
- Residual solvent extraction for injectable depot microspheres
Aseptic supercritical purification
Some fragile polymers can neither be purified by conventional methods nor terminally sterilized.
We took up this challenge for a client and achieved the world’s first aseptic supercritical fluid purification of a very challenging injectable medical polymer.
Sterile clinical trial materials were produced in compliance with the ISO 13408 standards and a full documentation and validation package, including process simulation tests, was delivered to our client.

